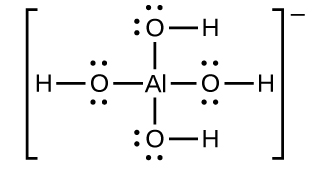
The Gibbs' energy for the decomposition of Al2O3 at 500^oC is as follows: 23Al2O3→ 43 Al + O2; ΔrG = + 960 kJmol^-1 The potential difference needed for the electrolytic reduction of

Al+O2=Al2O3 balance the equation @mydocumentary838. al+o2=al2o3 balance the chemical equation. - YouTube
What is the chemical formula for the compound formed between Al and O: Al3O2, AlO, Al2O, Al2O3, or AlO2? - Quora

Metal–support cooperation in Al(PO3)3-supported platinum nanoparticles for the selective hydrogenolysis of phenols to arenes | Nature Catalysis

Chemistry - Chemical Bonding (18 of 35) Lewis Structures for Ionic Comp - Aluminum Oxide - Al2O3 - YouTube








![LiAlH[Ot-Bu]3 For The Reduction of Acid Halides To Aldehydes – Master Organic Chemistry LiAlH[Ot-Bu]3 For The Reduction of Acid Halides To Aldehydes – Master Organic Chemistry](https://cdn.masterorganicchemistry.com/wp-content/uploads/2022/11/2-example-of-redution-of-acid-chlorides-to-aldehydes-using-lithium-tri-t-butoxy-aluminum-hydride.gif)