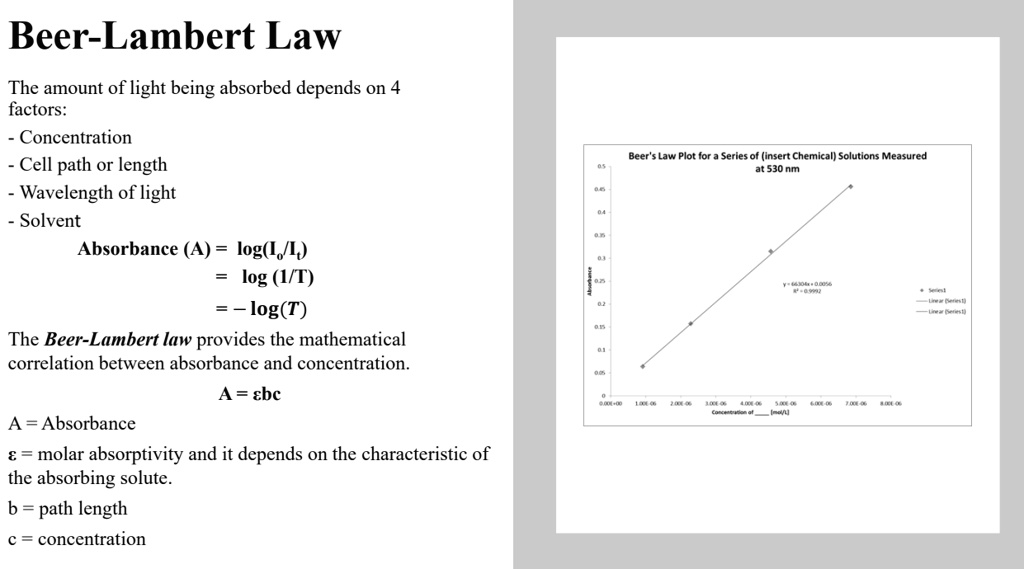
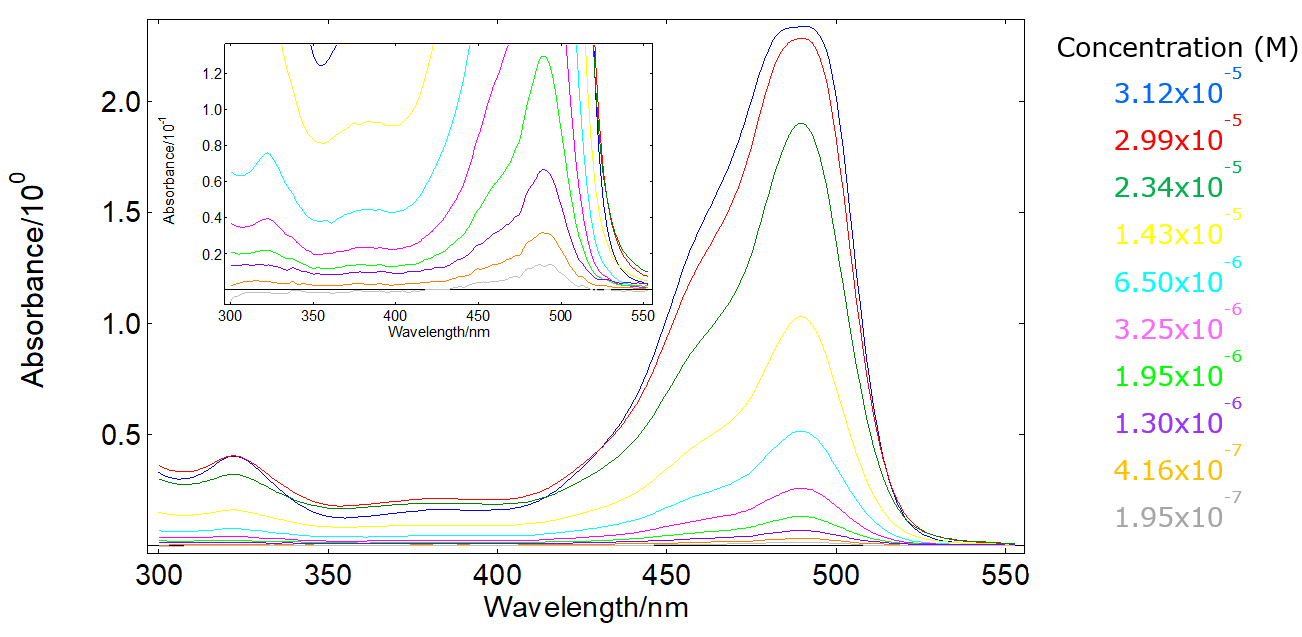
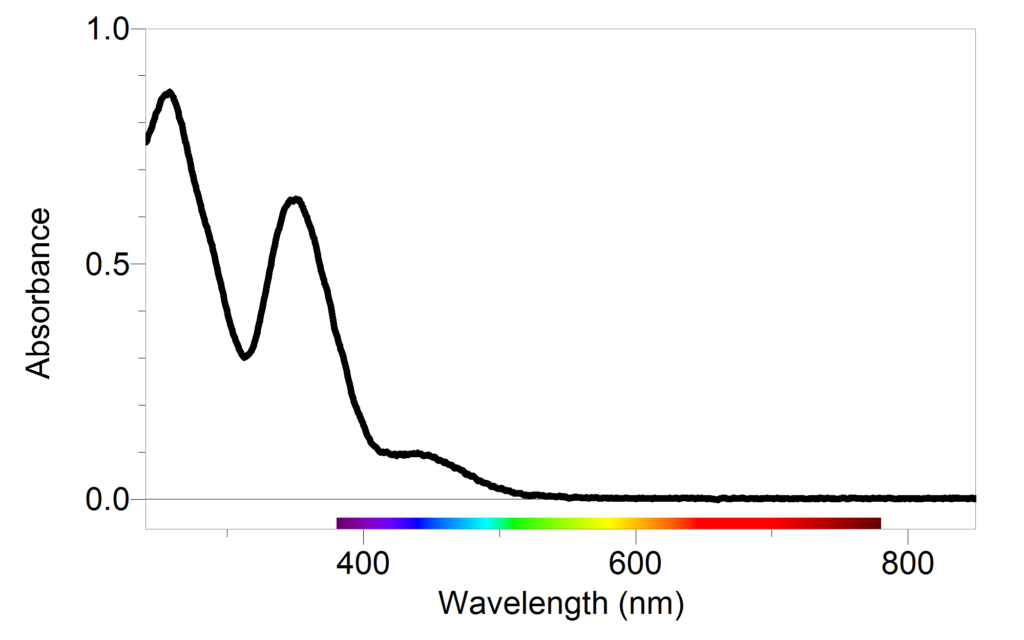
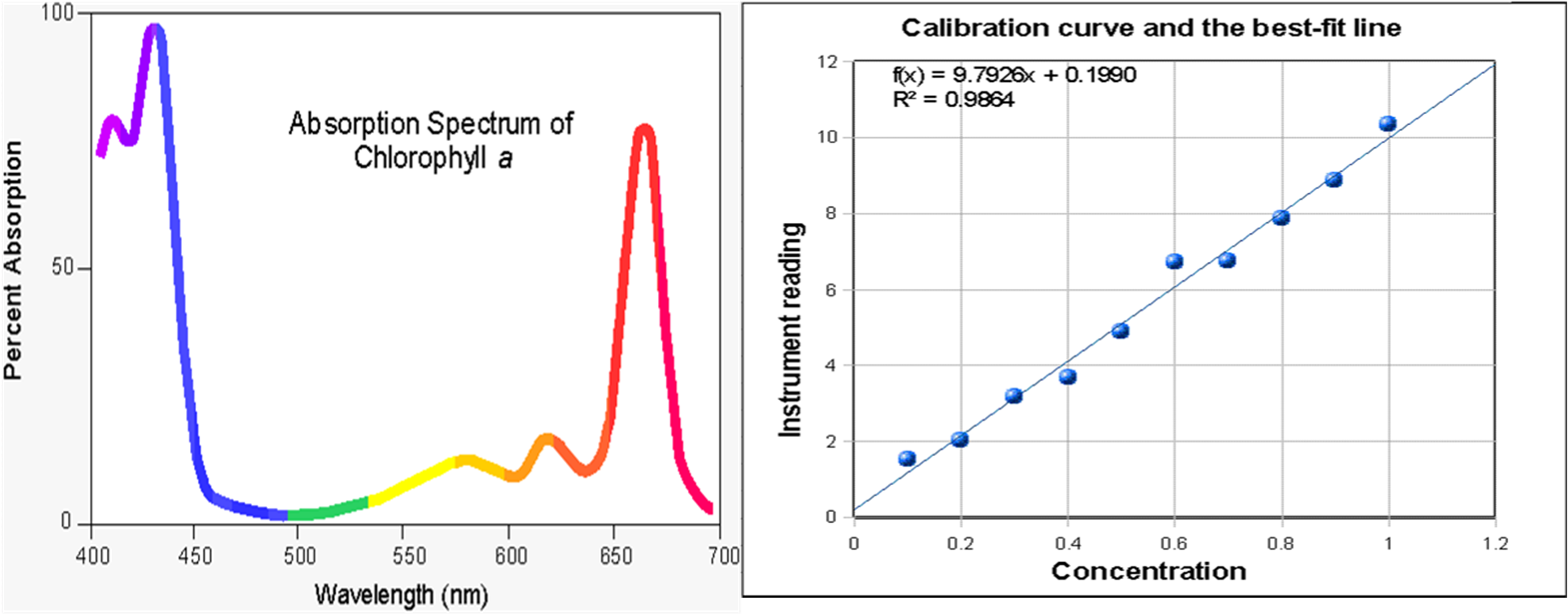
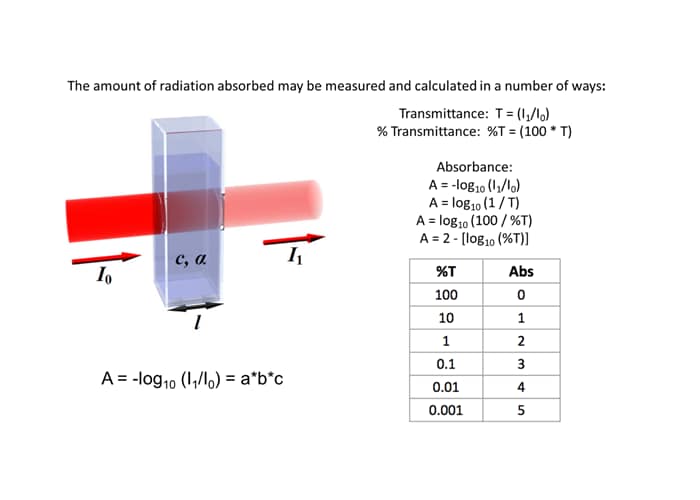
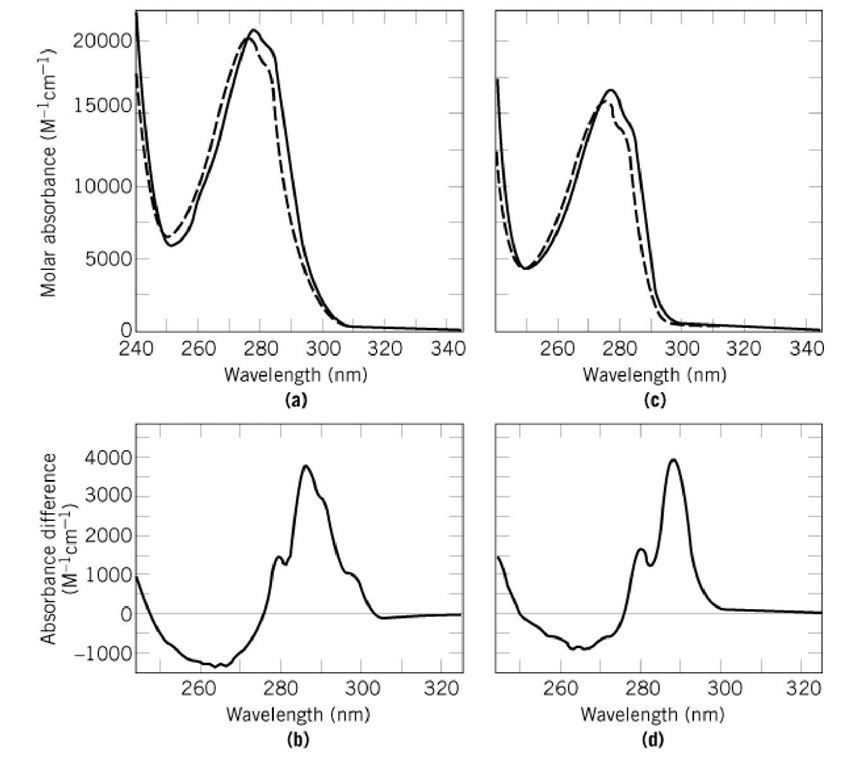
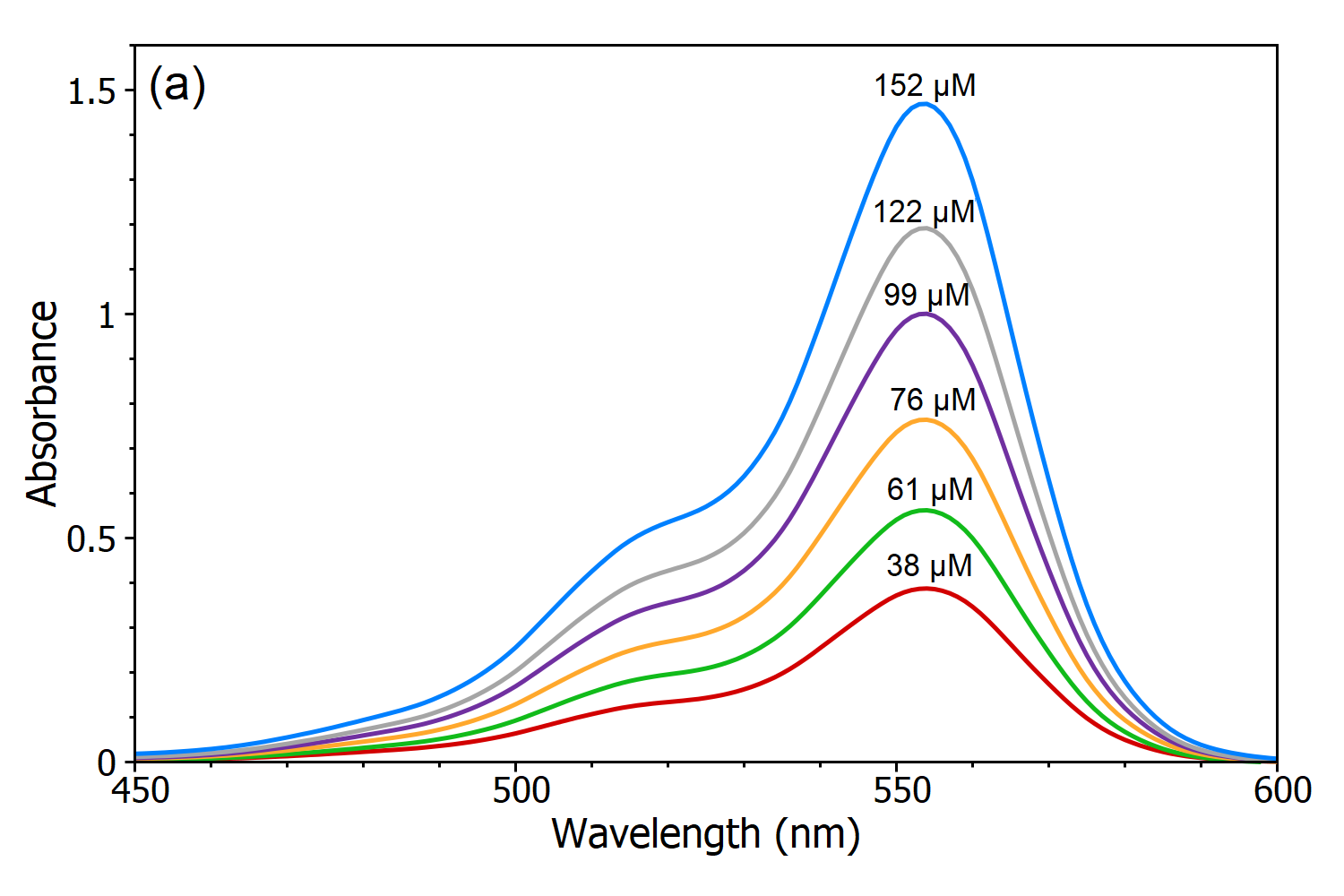
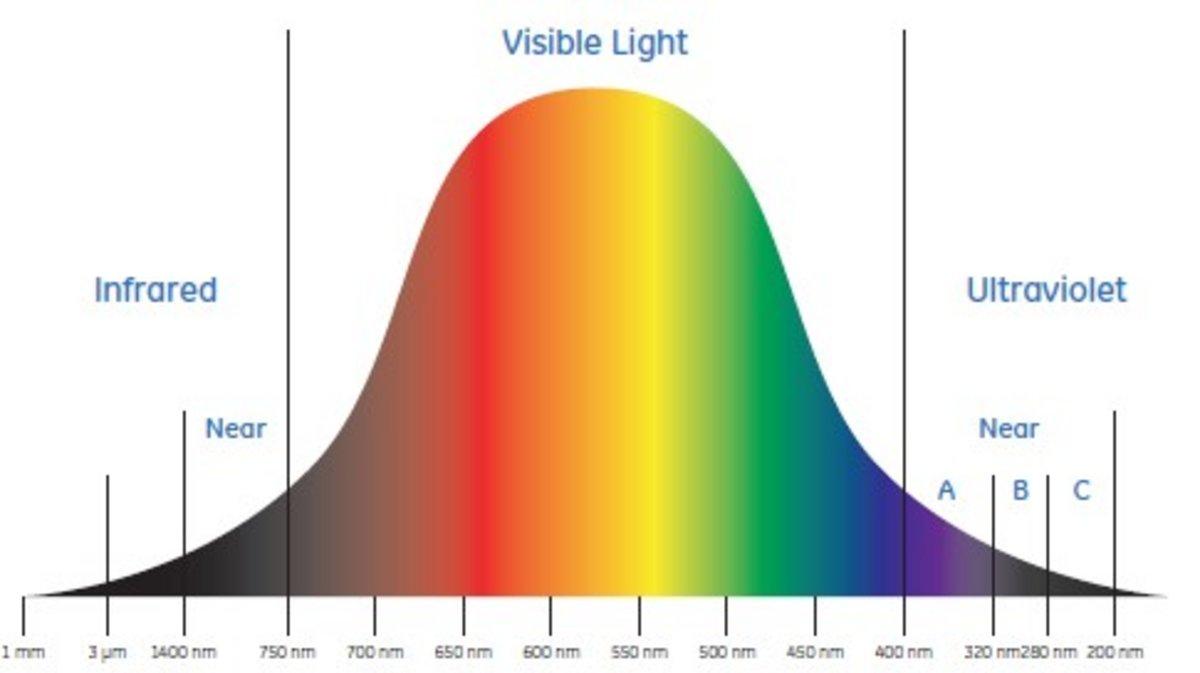
Why is it advisable to use the maximal wavelength for an absorbance peak when measuring the absorbance to determine the concentration of a compound? - Quora

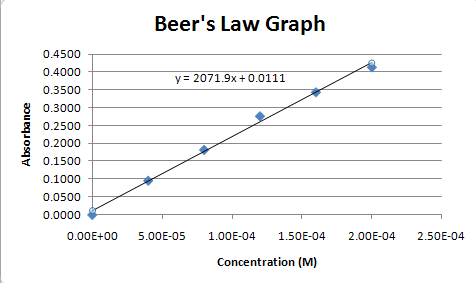
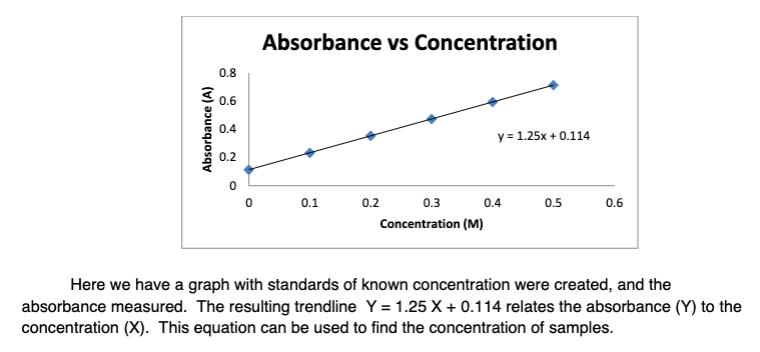
Use the data to plot a graph of Absorbance versus Concentration. Determine the concentration of the dye which would correspond to an absorbance of 0.140. What does Beer's Law say about the

Absorbance measurements (in absorbance units, A.U.) vs. optical path... | Download Scientific Diagram

Typical absorbance (A) and reaction kinetic rate (B) for MB degradation... | Download Scientific Diagram

Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions